Life sciences companies must juggle simultaneous pressures—end-to-end material traceability demands, strict shipping conditions to protect time- and temperature-sensitive products, and stringent government regulations, to name a few. Pharmaceutical, biotech, and medical device manufacturers that rely on generic or disconnected software solutions can’t always keep pace, as data silos hinder decision-making, introduce compliance risks, and complicate scaling at every stage, from initial research to commercial manufacturing.
Fortunately, specialized life sciences ERP systems integrate all departments—R&D, laboratories, clinical trials, manufacturing, quality management, supply chain, and financial operations—into one platform that provides real-time visibility and maintains the rigorous compliance standards these businesses must satisfy.
What Is Life Sciences ERP?
Life sciences ERP systems are integrated software platforms designed for research, pharmaceutical, biotech, diagnostics, and medical device companies. These systems unify operations—R&D, manufacturing, quality management, financial reporting—in a single platform that offers centralized data management, regulatory compliance, and end-to-end traceability.
Unlike generic ERP solutions, life sciences ERP systems are built for industry-specific requirements, including batch tracking, serialization, and integration with laboratory information management systems (LIMS) and quality management systems (QMS). This specialization allows life sciences companies to adhere to the rigorous documentation and traceability standards mandated by regulatory agencies, including the US Food and Drug Administration (FDA) regulation 21 CFR Part 11 that covers electronic records, while supporting efficient and scalable operations throughout the product lifecycle.
Key Takeaways
- Life sciences ERP systems integrate R&D, manufacturing, supply chain, quality, and finance operations to provide real-time visibility and maintain compliance.
- Material and batch traceability features document the complete journey of products from raw material sourcing through distribution, simplifying recalls and audits.
- ERP solutions automate workflows, allowing life sciences companies to scale from pre-revenue startups to global commercial operations without major system overhauls.
- ERP selection calls for businesses to evaluate their current and future needs, including customization, team involvement, regulatory requirements, and integration with existing systems.
ERP for Life Sciences Explained
Life sciences operations face unique risks and requirements that generic business software can’t always address, often stemming from the need for full visibility into global supply chains that contain sensitive customer and patient data. According to 2025 McKinsey research, maximizing ERP value for life sciences businesses means focusing on three approaches: using technology to transform processes, rather than simply digitizing existing ones; adopting a holistic value chain perspective; and integrating solutions that foster collaboration. An effective ERP system should address the following unique considerations of the life sciences industry:
- Material traceability: Each component in every life sciences product must be tracked for quick identification and responsiveness in the event of recalls or quality issues. ERP systems assign unique batch or lot numbers to each product, creating a digital record of every step of the product lifecycle and accelerating both internal and regulatory audits by automatically generating detailed production histories for each product line as needed.
- Complicated supply chain: ERP systems provide a comprehensive view of global operations through real-time shipment tracking and certification verification. Integration with Internet of Things devices and supplier management systems help life sciences companies monitor conditions and quickly pivot operations during delays through automated procurement processes or production schedule adjustments.
- Regulatory requirements: Life sciences companies, especially global businesses, must comply with stringent and complex compliance standards, such as the FDA Current Good Manufacturing Practice (CGMP) rules or the European Medicines Agency (EMA) Annex 11 regulations pertaining to the use of computer systems for medicinal products. Industry-specific ERP systems help companies demonstrate compliance through such features as electronic signatures and robust documentation throughout the manufacturing process.
- Data sensitivity: Clinical and patient data are highly sensitive and mandate robust security protocols. Life sciences ERP systems use encryption, role-based access controls, and automated approval workflows to safeguard data without limiting authorized users’ access to information they need.
- Systems integration: Many life sciences companies rely on specialized software for laboratory management, quality control, clinical trials, research, supply chain management, financials, and more. An effective ERP solution must integrate all existing systems under one management platform to eliminate data gaps, conversion errors between departments, and duplicate entries.
Benefits of Life Sciences ERP Solutions
Specialized life sciences ERP systems generate measurable and scalable operational improvements. Integration between laboratory systems and financial platforms minimizes errors and inconsistent documentation. At the same time, controlled access lets R&D, manufacturing, and quality teams securely share formulation details and production records without compromising sensitive clinical data. Life sciences ERP systems deliver wide-ranging benefits, including the following, which demonstrate particular impact in seven key areas:
- Efficiency gains: Automation and integration minimize manual data entry, decrease errors, and accelerate workflows. Laboratory teams can automatically transfer batch testing results to manufacturing systems; quality teams can route deviations through automated approval processes; and production staff can access validated formulas without manual transcription—leading to higher productivity and faster responses when problems surface.
- Regulatory adherence: Built-in compliance features help life sciences companies meet FDA, EMA, and international regulations through automated recordkeeping and compliant electronic report generation. These features lower both audit preparation time and the risk of incurring noncompliance penalties that can delay or halt production and harm profitability.
- Unified data management: ERP platforms integrate all business functions into a single system, providing data management with real-time performance visibility while eliminating information silos and translation errors. Real-time dashboards give executives and managers immediate access to key performance indicators, such as batch yields, material expiration rates, quality deviation trends, and clinical trial enrollment metrics, thereby eliminating the need for manual reports.
- Secured information: Centralized data storage uses strong security protocols to prevent unauthorized personnel from accessing or modifying critical information and to maintain appropriate access for research, regulatory, and manufacturing teams. Advanced encryption, secure cloud storage, and regular security updates safeguard proprietary formulations, clinical trial data, patient identifiers (in accordance with HIPAA requirements), and intellectual property.
- More accurate reporting: Data integration tools automatically consolidate and update information from different departments, giving decision-makers immediate insights into material inventory relative to production schedules, cold chain compliance for biologics shipments, and deviation rates by product line. This data helps companies quickly handle raw material shortages or batch failures, informing decisions with accurate analytics, rather than outdated reports or guesswork.
- Cross-functional coordination: When everyone works from a unified data source, teams can share information and coordinate efforts regardless of department or location. For example, R&D teams can transfer validated manufacturing processes to production sites, quality teams can alert supply chain managers to new supplier audit findings, and regulatory managers can access batch records when submitting records to health authorities.
- Financial visibility: Unified financial operations connect accounting with manufacturing, inventory, and sales to provide an up-to-date picture of financial health that helps executives when they’re planning research investments and commercial expansions. ERP for life sciences also improves cash flow visibility, thanks to pharmaceutical revenue recognition that provides automatic adjustments for rebates and chargebacks, R&D trackers that allocate spending by project and clinical trial phase, and bookkeeping processes that automatically account for international regulatory fees.
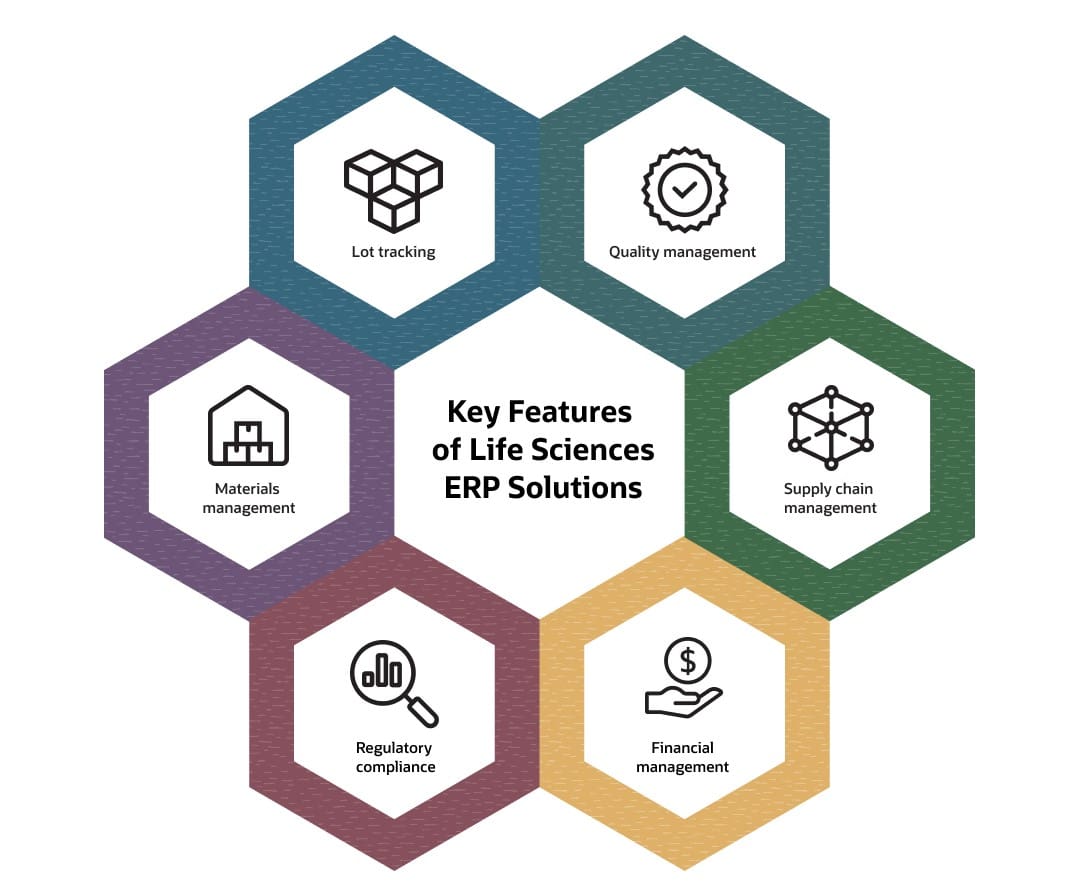
Key Features of Life Sciences ERP Solutions
Life sciences companies look for specialized features that exceed capabilities of out-of-the-box software. For example, companies must maintain granular traceability logs and documentation for sensitive ingredients and components and then integrate this traceability directly with production workflows to prevent non-conforming materials from entering batches. This level of quality management—far beyond the purview of standard business controls—must extend to every link in the supply chain, including temperature-controlled shipping conditions and financial transparency across global distribution networks. For many companies, the ERP for life sciences features outlined below aren’t optional—they’re required by regulators to maintain consistent product quality and safety.
-
Lot Tracking
Lot tracking automatically generates unique lot and serial numbers for every batch, supporting comprehensive inventory management and visibility from production through distribution. The system assigns each batch a unique identifier, allowing companies to quickly pinpoint affected products along the supply chain if recalls become necessary. Physical traceability features include integrated barcode and RFID scanners, as well as analytics tools that track shelf life and assess trends to support and monitor the impact of improvement initiatives.
-
Quality Management
Quality management enforces standards at every stage of the product lifecycle by controlling materials, equipment, and personnel throughout manufacturing, processing, and shipping. When ingredients fall outside accepted specifications, the system alerts quality control teams to remove faulty materials or products. Integrated quality control features automate corrective and preventive actions and facilitate data exchange among ERP, manufacturing, QMS, and LIMS systems to provide real-time documentation for inspections to lessen the risk of subpar product batches reaching the market.
-
Material Management
Advanced inventory controls include strict material expiration dates, storage condition monitoring (temperature, pressure, and humidity), and detailed recordkeeping of item attributes and measurement units. Materials tracking capabilities use integrated demand forecasts, real-time utilization rates, and automated reorder thresholds to optimize stock levels. This proactive inventory approach lowers the chances of using expired materials, yet doesn’t sacrifice the need for sufficient supply levels to meet market needs.
-
Supply Chain Management
Supply chain management tools provide end-to-end traceability spanning sourcing to final delivery. They track supplier certifications, monitor shipment timelines, and verify that shipments meet necessary conditions throughout transport. ERP for life sciences platforms automate procurement planning, execution, and supplier management to make sure production materials are on hand when needed—even critical components with long lead times. Supply chain management also supports production planning, complex shipping requirements for sensitive materials, and real-time demand forecasting, thus preventing both shortages that lead to lost revenue and overstocks that tie up working capital and increase carrying costs.
-
Regulatory Compliance
Compliance management modules encompass detailed digital logs, electronic approvals, and automated tracking and documentation, all serving to demonstrate adherence to FDA, EMA, and other relevant standards. ERP systems support electronic records and signatures that meet FDA 21 CFR Part 11 requirements and uphold good manufacturing and laboratory practices. The result is shorter audit preparation time and less risk of regulatory penalties, especially when combined with automated updates that help organizations stay current with changing requirements.
-
Financial Management
Financial management capabilities connect accounting with research, manufacturing, and sales data to give finance teams immediate visibility into project and clinical-trial spending, investment returns, product profitability, and administrative overhead. Life sciences ERP systems give companies scalable financial tools to generate financial reports and that recognize revenue from multiple entities or global business units. With 56% of biopharma executives surveyed in Deloitte’s “2025 Life Sciences Outlook” report planning to rethink their R&D strategies, and 40% emphasizing the need to improve R&D productivity to counter declining returns, the real-time visibility into research spending and project performance provided by ERP systems becomes a valuable tool for businesses to maximize ROI by supporting data-driven decisions on resource investment and allocation.
Key Features of Life Sciences ERP Solutions
6 Questions to Help You Choose the Right Life Sciences ERP
Because every ERP system is unique, selecting the right one warrants careful evaluation of your business’s operations and how the system will handle your company’s specific workflows and projects. Potential ERP vendors should have life sciences expertise and be well-suited for the intricacies of the industry, verified by hands-on demos and peer reviews from businesses in similar fields. The six questions below can help decision-makers assess whether a platform can support current requirements and accommodate future growth in line with regulatory standards and operational priorities, including planned R&D cycles and product expansions:
- How customizable will you need your ERP to be? Assess whether existing operations will necessitate extensive customization or be fine with industry-standard configurations. Be aware that standard templates can simplify implementation and ongoing updates but will likely mean that staff will have to adapt their workflows, which can slow down adoption rates and defer ROI.
- Which teams will need to be involved in helping you choose an ERP system? Selecting an ERP solution should involve multiple stakeholders, including quality assurance teams to identify compliance gaps, manufacturing personnel to test production exceptions, IT staff for technical needs, finance departments for reporting and investor relations, R&D leaders for grant-tracking and IP management, and frontline users for day-to-day workflows. Including diverse representatives in the selection process helps implementation teams address pain points that leadership may overlook and gains buy-in from the teams that will directly work with the system.
- What support and training will your end users need? ERP systems often come with steep learning curves, especially for companies with long-established legacy systems. McKinsey’s 2025 research found that nearly 70% of ERP implementations fail to meet expectations, often because of the company’s implementation approach, rather than technical limitations. Evaluate each system’s training programs, documentation quality, and interface complexity relative to staff technical proficiency and available training time. And don’t forget to review vendors’ implementation policies and ongoing technical support.
- How scalable is the ERP system? ERP platforms should grow alongside a company’s trajectory without the need for major replacements. The system should scale to support operations at every stage, including pre-revenue research project management, clinical trials, and complete commercial manufacturing, with modules that can adapt to increased users, SKUs, facilities, business units, and regional jurisdictions.
- What compliance and security standards will you need to follow? Identify all regulatory requirements for each market your company operates in—or plans to enter—including FDA, HIPAA, EMA, and any country-specific rules. The right life sciences ERP should support all necessary compliance protocols, including data integrity controls and audit requirements, with documentation capabilities that can adapt to evolving standards.
- What other systems need to be integrated into your ERP? Map out all specialized software currently in use for laboratory data, quality control, real-time production monitoring, and clinical trial management. Determine which software the ERP system will replace and which will be integrated with the new platform. Many ERP solutions, especially those with cloud architecture and robust APIs, can exchange data among systems to eliminate information silos that introduce inaccuracies and delays when making decisions and generating reports.
How NetSuite’s All-in-One ERP Empowers Life Sciences Companies
Life sciences companies struggle with the presence of disconnected systems that may lead to compliance risks, limited visibility, and scaling challenges at every stage of the product lifecycle. NetSuite Life Sciences ERP replaces disjointed legacy software with a unified cloud platform that delivers complete visibility with end-to-end lot tracking and serialization. Native quality management capabilities embedded throughout production help life sciences businesses maintain product standards and support regulatory compliance in all international jurisdictions. Meanwhile, project-based accounting automates financial management for all sales channels and business units, and product lifecycle management tools track R&D projects, clinical trial phases, and research programs.
Track Each Project With NetSuite
For life science companies choosing an ERP system, the right platform must address multiple industry-specific challenges, including regulatory compliance, material and product traceability, integration with specialized systems, and scalability. By consolidating R&D, manufacturing, quality management, and finance data into a single platform, businesses can maintain audit-ready records, track materials throughout global supply chains, and coordinate projects. Life sciences companies that invest in the right ERP system position themselves to respond quickly to market demands and support sustainable growth.
ERP For Life Sciences FAQs
What is one of the most critical requirements for a life science ERP?
Regulatory compliance is a critical requirement, as life sciences companies must adhere to strict government and industry regulations. ERP systems for life sciences provide built-in compliance features, including electronic signatures, comprehensive audit trails, and validation-ready processes that make sure records are accurate and secure.
Who are the people involved in the ERP implementation?
Successful ERP implementation requires a cross-functional team that should include executive leadership for strategic direction, IT personnel for technical requirements, finance teams to verify accounting features, quality assurance specialists to confirm compliance capabilities, operations staff to configure production workflows, and end users from each department to test the system and promote buy-in.
How long does it take to implement an ERP?
Implementation timelines vary based on company size, system complexity, and desired customization, with prebuilt systems taking as little as a few months to deploy and more complex implementations taking longer, sometimes spanning multiple years. Factors influencing this timeline include data migration complexity, integration requirements with existing systems, compliance documentation needs, and the company’s change management capabilities.








